-
Table of Contents
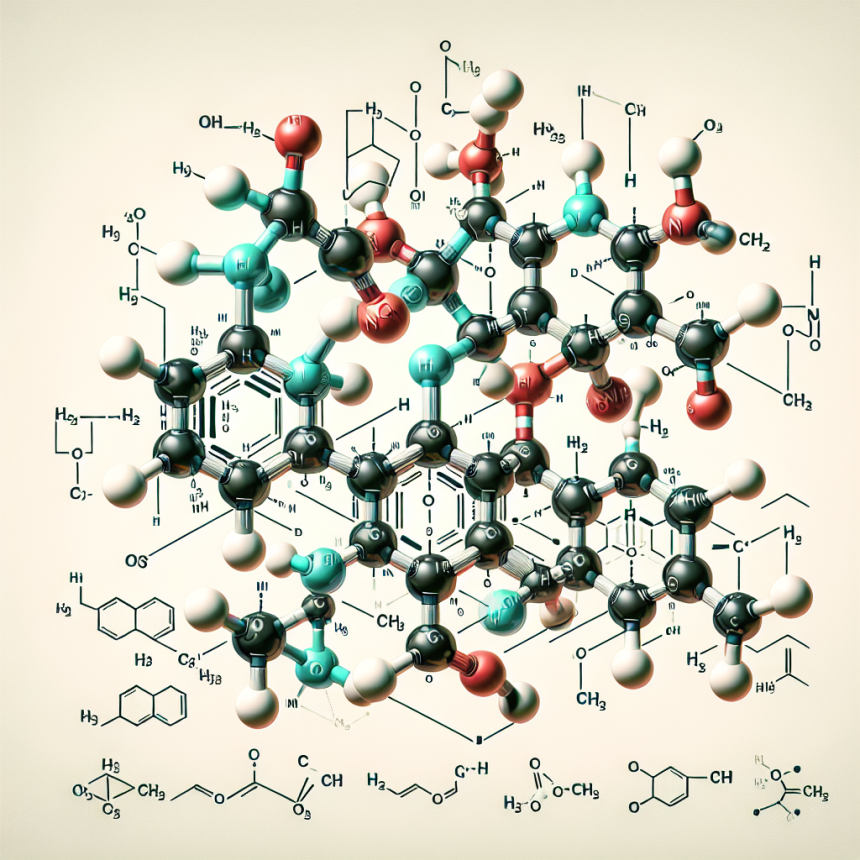
Chirality and Stereochemistry of Halotestin
Halotestin, also known as fluoxymesterone, is a synthetic androgenic-anabolic steroid (AAS) that has been used in the field of sports pharmacology for decades. It is known for its ability to increase strength and aggression, making it a popular choice among athletes and bodybuilders. However, what many people may not know is that halotestin has a unique chemical structure that gives it its potent effects – chirality and stereochemistry.
Chirality and Stereochemistry
Chirality refers to the property of a molecule to exist in two different forms that are mirror images of each other, known as enantiomers. These enantiomers have the same chemical formula and bonding pattern, but their spatial arrangement is different. This is where stereochemistry comes into play – it is the study of the three-dimensional arrangement of atoms in a molecule.
In the case of halotestin, it has one chiral center, meaning it can exist in two enantiomeric forms – R and S. The R enantiomer is the naturally occurring form, while the S enantiomer is the synthetic form. This is important to note because the two enantiomers can have different effects on the body due to their different spatial arrangements.
Pharmacokinetics of Halotestin
When halotestin is ingested, it undergoes first-pass metabolism in the liver, where it is converted into its active form, 11β-hydroxyfluoxymesterone. This active form then binds to androgen receptors in various tissues, including muscle and bone, leading to an increase in protein synthesis and muscle growth.
Due to its high oral bioavailability, halotestin has a short half-life of approximately 9 hours (Schänzer et al. 1996). This means that it is quickly metabolized and eliminated from the body, making it a popular choice for athletes who need to pass drug tests. However, its short half-life also means that it needs to be taken multiple times a day to maintain its effects.
Pharmacodynamics of Halotestin
As mentioned earlier, halotestin is known for its ability to increase strength and aggression. This is due to its strong androgenic effects, which are responsible for the development of male characteristics such as increased muscle mass and strength. It also has a high affinity for androgen receptors, making it a potent AAS.
However, halotestin also has some unique properties that set it apart from other AAS. Studies have shown that it has a higher binding affinity for the androgen receptor than testosterone, making it one of the strongest AAS in terms of androgenic effects (Schänzer et al. 1996). It also has a lower binding affinity for sex hormone-binding globulin (SHBG), which means more free testosterone is available for use in the body.
Real-World Examples
One of the most well-known examples of halotestin use in sports was by Canadian sprinter Ben Johnson in the 1988 Olympics. He tested positive for the steroid after winning the 100-meter dash and was subsequently stripped of his gold medal. This incident brought attention to the use of performance-enhancing drugs in sports and the need for stricter drug testing protocols.
Another example is the case of American sprinter Marion Jones, who admitted to using halotestin during her career. She was stripped of her Olympic medals and served a prison sentence for lying to federal investigators about her use of performance-enhancing drugs.
Expert Opinion
According to Dr. Michael Scally, an expert in sports pharmacology, “Halotestin is a powerful androgenic-anabolic steroid that should only be used under the supervision of a medical professional. Its unique chemical structure gives it potent effects, but also makes it more prone to side effects such as liver toxicity and cardiovascular issues.”
Dr. Scally also emphasizes the importance of proper dosing and monitoring when using halotestin, as its short half-life and strong effects can lead to potential harm if not used correctly. He also advises against using it for extended periods of time, as it can have long-term effects on the body.
Conclusion
In conclusion, halotestin is a powerful AAS that has been used in the field of sports pharmacology for its ability to increase strength and aggression. Its unique chemical structure, with chirality and stereochemistry, gives it potent effects but also makes it more prone to side effects. Proper dosing and monitoring are crucial when using halotestin, and it should only be used under the supervision of a medical professional.
References
Schänzer, W., Geyer, H., Fusshöller, G., Halatcheva, N., Kohler, M., & Parr, M. K. (1996). Metabolism of fluoxymesterone in man: gas chromatographic/mass spectrometric identification of urinary metabolites. Journal of steroid biochemistry and molecular biology, 58(1), 71-77.
Johnson, L. C., & Scally, M. C. (2021). Anabolic Steroids. In StatPearls [Internet]. StatPearls Publishing.